Extracellular vesicles (EVs) are highly involved in the progression of diverse diseases. Considering that, targeting EV biogenesis is a potential strategy for the treatment of the related diseases, which urges an improved understanding of EV biogenesis. During the biogenesis of EV subset, exosomes, invagination of the plasma membranes forms early endosomes which mature into late endosomes and multivesicular bodies (MVBs) eventually. MVBs fuse with not only the plasma membranes to release EVs but also lysosomes for degradation. Rab7 participates in the lysosomal targeting of MVBs. However, the proteins on MVB that directly bind Rab7, causing MVB recruitment of Rab7 remain unidentified.
The research team led by Prof. CAI Zhijian from the Zhejiang University School of Medicine discovered the novel mechanism of EV biogenesis. The findings were published in an article titled “Neddylation of Coro1a determines the fate of multivesicular bodies and biogenesis of extracellular vesicles” in the Journal of Extracellular Vesicles on October 8.
“Ubiquitin and ubiquitin-like modifications contribute to the regulation EV biogenesis,” said CAI, “Neddylation refers to the covalent attachment of the ubiquitin-like protein NEDD8 to a target protein, which is related to neurodegenerative diseases, cancer and infection. Whether neddylation modification can also regulate EV biogenesis has to be further explored.”
In this study, researchers found that Coro1a would undergo neddylation modification at K233 by TRIM4. Neddylated Coro1a is associated with the MVB membrane and facilitates MVB recruitment and activation of Rab7 by directly binding Rab7. Subsequently, MVBs are targeted to lysosomes for degradation in a Rab7-dependent manner, leading to reduced EV secretion.
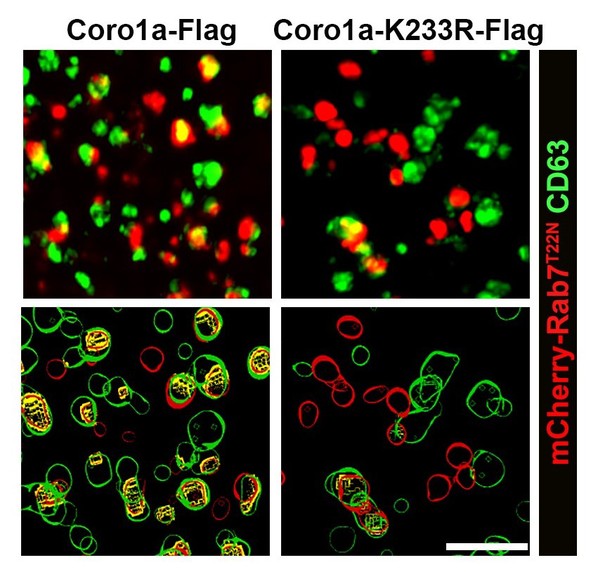
NEDD8-Coro1a mediates the recruitment of Rab7 to MVBs
Furthermore, researchers found MC38-Coro1a-K233R tumour-bearing mice exhibited faster tumour growth and shorter survival than MC38-Coro1a tumourbearing mice, inhibiting that a decrease in neddylated Coro1a enhances the production of tumour EVs, thereby promoting tumour progression, indicating that neddylated Coro1a is an ideal target for the regulation of EV biogenesis.
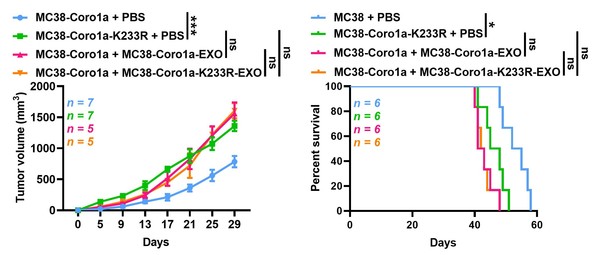
Inhibition of Coro1a neddylation promotes tumour progression by increasing EV secretion
“Our research identified a novel substrate of neddylation and revealed an unknown mechanism for MVB recruitment of Rab7, thus providing new insight into the regulation of EV biogenesis,” said Prof. CAI.
Source:CAI Zhijian‘s Lab